|
8/28/2023 0 Comments Compound molecular geometry table
What is difference between electron proton and neutron?.What are the characteristics of the alkaline earth metals?.What is the physical properties of alkali metals?.What is the Electronic configuration of iron and iron ions.Bohr's Atomic Model And Its Limitations.Distinguishing Tests For Organic Compounds.Properties and Molecular Structure of Oxygen.Nucleophilicity and basicity difference.what is stability order of carbocations.Explain all chemical Test for alcohol and phenols.what are the list of tests for Aldehydes and Ketones.How shapes of molecules are related to the type of hybridization.How Ionic bond forms ? What are the properties.Table of valence shell electron pair repulsion theory VSEPR Shape of Molecules/Ions having only bond Pairs of Electrons 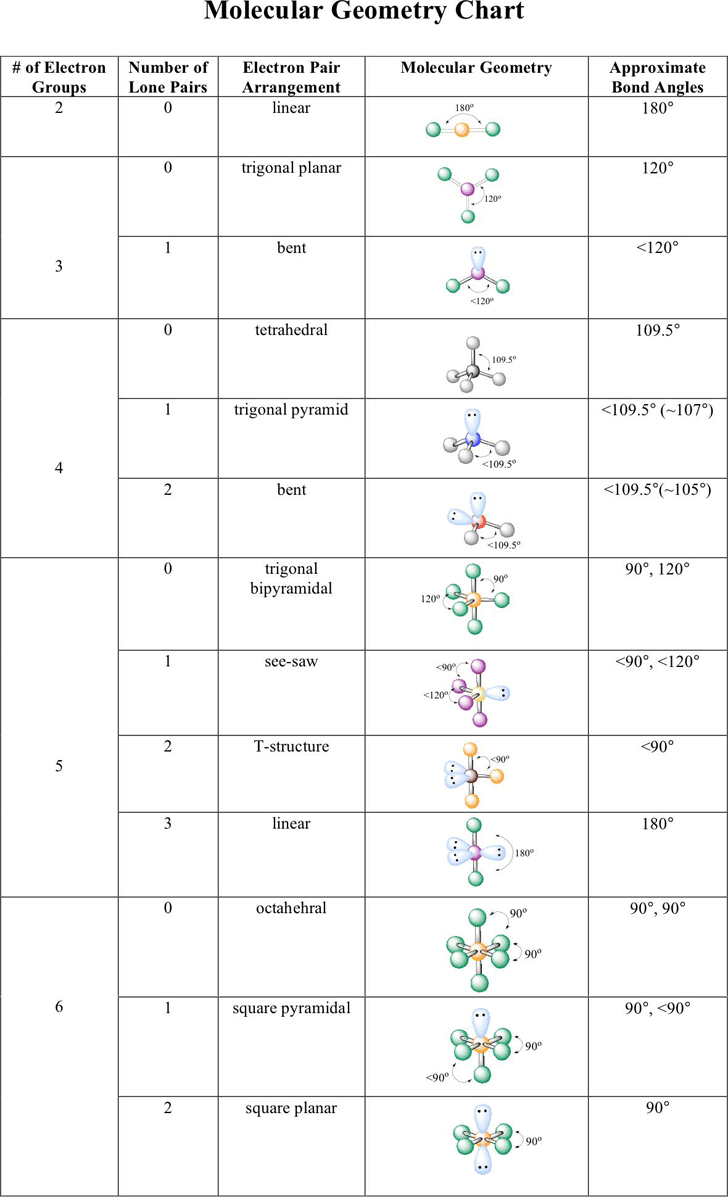
Geometry corresponding to number of bond pairs and lone pairs are given in the following table. If there are only bond pairs in the valence shell around the central atom it has regular geometry but when there are lone pairs also in the valence shell, the geometry becomes irregular due to increase in force of repulsion. Thus the Molecular geometry is determined by the number and nature of pairs of electrons surrounding the central atom. The repulsive forces between various types of electron pairs are in the order 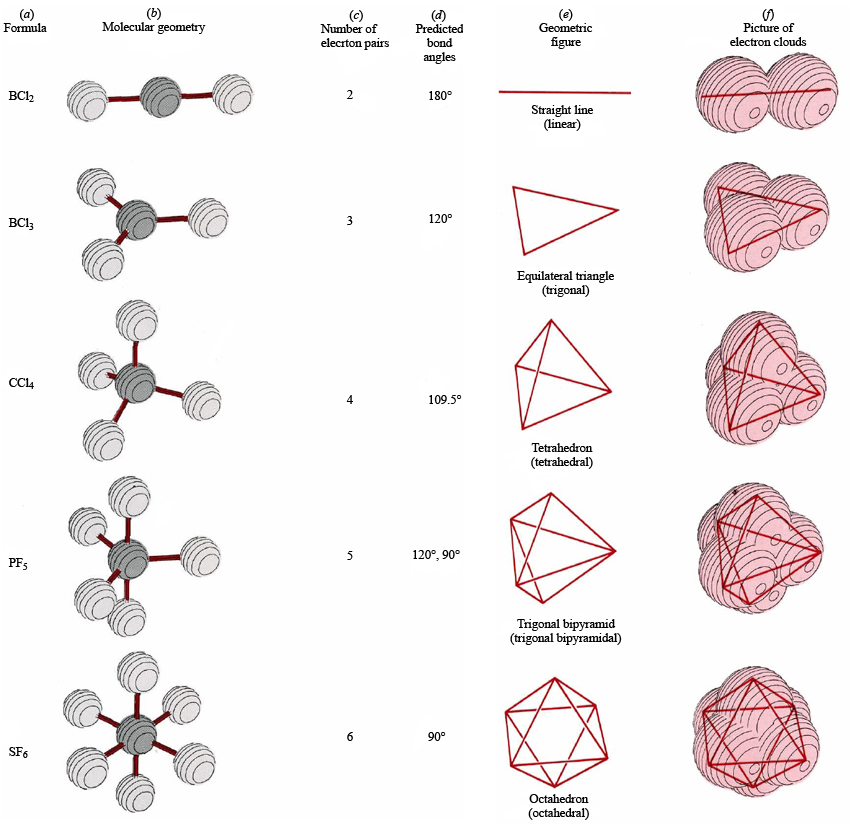
These electron pairs extend a repulsion force on one another and so lie as far apart from one another as possible so that the force of repulsions are minimized. There is a central atom, which is surrounded by bonded electron pairs or lone pairs of electron and shared pair of electrons in its valence shell. This theory is known as valence shell electron pair repulsion theory, which determines the shape and molecular geometry in a polyatomic molecule. For molecules that contain only single bonds.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
